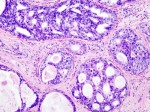
10 Newly-Defined Molecular Types of Breast Cancer in Nature, and a Dream
The 10 molecular BC categories bear prognostic (survival) information and, based on their distinct mutations and gene expression patterns, potential targets for novel drugs….I wonder if, in a few years, some breast cancers might be treated without surgery.